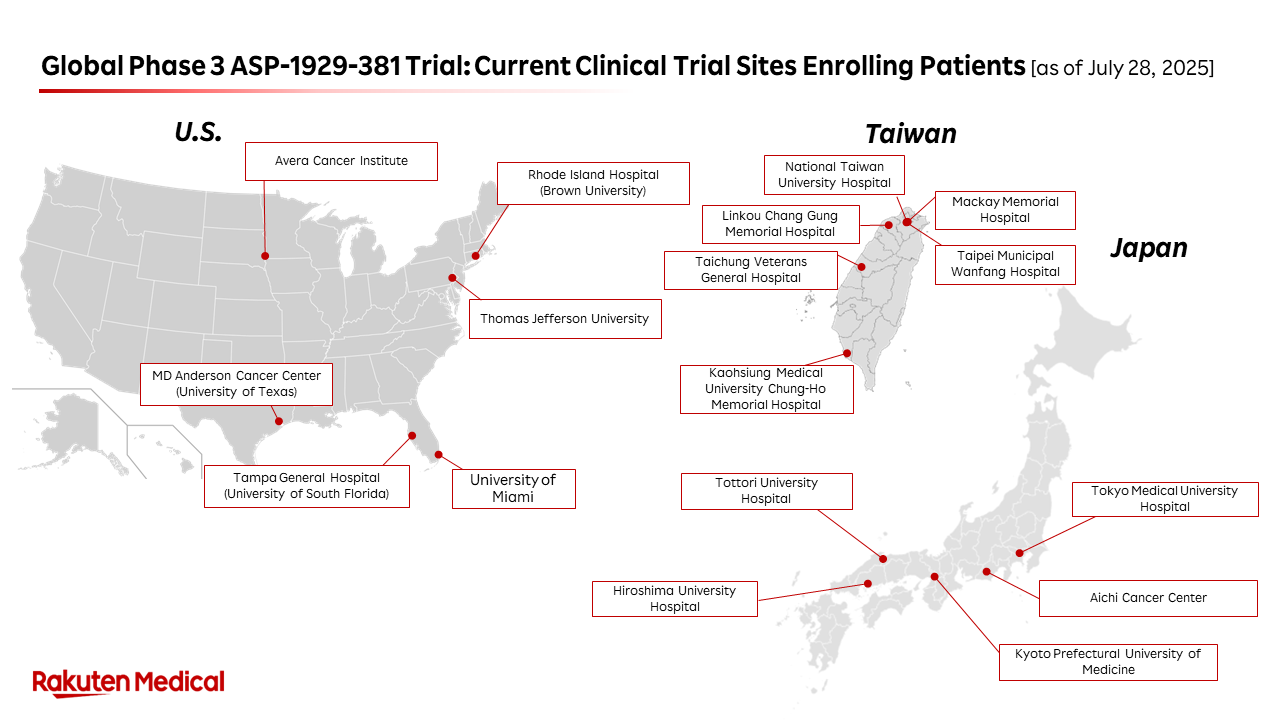
- Over 15 clinical sites in the U.S., Taiwan and now Japan are actively recruiting patients
- Planned site expansion into Eastern Europe with additional clinical trial sites in Ukraine and Poland
San Diego, CA — Rakuten Medical, Inc., a global biotechnology company developing and commercializing Alluminox™ platform-based photoimmunotherapy, today announced the expansion of its ongoing global Phase 3 trial, ASP-1929-381, to include patient enrollment in Japan. The trial is evaluating ASP-1929 photoimmunotherapy in combination with pembrolizumab as a first-line therapy for patients with recurrent head and neck squamous cell carcinoma (HNSCC) (Protocol number: ASP-1929-381 / Acronym: ECLIPSE / ClinicalTrials.gov Identifier: NCT06699212).
Currently, over 15 clinical sites across the United States, Taiwan and Japan are actively recruiting patients. Additional sites are expected to be activated in these regions. To further accelerate recruitment, Rakuten Medical also plans to expand the study to Eastern Europe, with new sites to be activated in Ukraine and Poland in the near future.
This global Phase 3 trial builds upon encouraging data from an interim evaluation (data cut-off: August 31, 2023) of 19 patients enrolled in the HNSCC cohort of a Phase 1b/2 study (ASP-1929-181/ClinicalTrials.gov Identifier: NCT04305795). That study evaluated ASP-1929 photoimmunotherapy in combination with anti-PD-1 including pembrolizumab in patients with recurrent or metastatic HNSCC. The data showed an estimated 24-month survival rate of 52.4%, with a median OS not yet reached, and a generally manageable safety profile.
“Our global Phase 3 study is making solid progress across the U.S., Taiwan, and Japan,” said Mickey Mikitani, Chief Executive Officer of Rakuten Medical. “To further accelerate enrollment, we are expanding our network of study sites and preparing to roll out new sites in Ukraine and Poland. Designed for widespread accessibility, our device is portable and cost-effective. We believe our therapy has the potential to make a meaningful contribution to societies worldwide, including those with emerging healthcare systems and considerable unmet needs in oncology.”
About ASP-1929-381 Study
The ASP-1929-381 study is a multi-regional multi-center, randomized, open-label Phase 3 trial, designed to assess the efficacy and safety of ASP-1929 photoimmunotherapy in combination with pembrolizumab as a first-line treatment for locoregional recurrent HNSCC without distant metastases. 412 patients globally will be randomized to either an experimental arm receiving ASP-1929 photoimmunotherapy in combination with pembrolizumab, or a control arm receiving the current pembrolizumab-based standard of care (SOC), where patients may receive pembrolizumab alone or in combination with chemotherapy according to the physician's choice.
The primary endpoint is Overall Survival (OS), with key secondary endpoints including Complete Response Rate (CRR) and Overall Response Rate (ORR).
Disclaimer: Rakuten Medical’s Alluminox™ platform-based photoimmunotherapy is investigational outside Japan.
About Rakuten Medical, Inc.
Rakuten Medical, Inc. is a global biotechnology company developing and commercializing Alluminox™ platform-based photoimmunotherapy, which, in pre-clinical studies, has been shown to induce rapid and selective cell killing. Rakuten Medical’s photoimmunotherapy is currently investigational outside Japan. Rakuten Medical is committed to its mission to conquer cancer by developing its pioneering treatments as quickly as possible to as many patients as possible all over the world. The company has offices in 5 countries/regions, including the United States, where it is headquartered, Japan, Taiwan, Switzerland and India. For more information, visit www.rakuten-med.com.
About Alluminox™ platform
The Alluminox™ platform is Rakuten Medical’s investigational technology platform that combines pharmaceuticals, medical devices, medical technology, and other peripheral technologies. Rakuten Medical is developing Alluminox platform-based photoimmunotherapy, which involves two key steps: 1) drug administration and 2) targeted illumination using medical devices. The drug component consists of a cell-targeting moiety conjugated to a light-activatable dye, such as IRDye® 700DX (IR700), that selectively binds to the surface of targeted cells, such as tumor cells. The device component consists of a light source that locally illuminates the targeted cells with red light (690nm) to transiently activate the drug. Rakuten Medical’s pre-clinical data have shown that this activation elicits rapid and selective necrosis of targeted cells through a biophysical process that compromises the membrane integrity of the targeted cells. Therapies developed on the Alluminox platform may also result in local and systemic innate and adaptive immune activation due to immunogenic cell death of the targeted tumor cells and/or the removal of targeted immunosuppressive cells within the tumor microenvironment. Photoimmunotherapy was originally developed by Dr. Hisataka Kobayashi and his team at the National Cancer Institute in the United States. Outside Japan, Rakuten Medical’s Alluminox platform-based photoimmunotherapy is investigational.
About ASP-1929
Rakuten Medical’s first pipeline drug developed on its Alluminox™ platform is ASP-1929, an antibody-dye conjugate comprised of the anti-EGFR antibody cetuximab and IRDye® 700DX, a light activatable dye. ASP-1929 binds to epidermal growth factor receptor (EGFR), a cancer antigen expressed in multiple types of solid tumors, including head and neck, breast, lung, colorectal, prostate and pancreatic cancers. After binding to cancer cells, ASP-1929 is locally activated by illumination with red light (690 nm), emitted by a laser device system to produce a photochemical reaction. This reaction is believed to cause damage to the membrane of cancer cells, leading to selective necrosis of cancer cells. In Japan, ASP-1929 received marketing approval from the Japanese Ministry of Health, Labor, and Welfare for unresectable locally advanced or recurrent head and neck cancer in September 2020, under the Sakigake Designation System and the Conditional Early Approval System. ASP-1929 photoimmunotherapy in combination with pembrolizumab is currently under investigation in a global Phase 3 clinical trial as a first-line therapy for recurrent head and neck cancer. Outside Japan, ASP-1929 has not yet been approved for commercial use by any regulatory authority.
